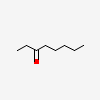
Gas chromatography-mass spectrometry ID'd the culprit as the volatile ketone 3-octanone.
See full article...
See full article...
Specially 3 days after the first episode of "The last of us"...Carnivorous fungi is a mind picture I could do without. Thanks.
It's actually used a flavouring agent and as a scent agent in products for human consumption. Gives things a "fruity, cheesy, mushroom odour", which makes sense.Wonder what 3-octanone from those mushrooms does in the human system? And since they’re volatile, I’m assuming cooking/sautéing the mushrooms lowers the toxicity profile?
Probably not much.Wonder what 3-octanone from those mushrooms does in the human system? And since they’re volatile, I’m assuming cooking/sautéing the mushrooms lowers the toxicity profile?
As a class Nemacitides aren't something you want to ingest (especially, I suppose, if you are a nematode). They seem to be fairly toxic.Wonder what 3-octanone from those mushrooms does in the human system? And since they’re volatile, I’m assuming cooking/sautéing the mushrooms lowers the toxicity profile?
You may be joking, but there are some concerns that as the climate warms up fungi will adapt to higher temperatures and eventually survive at human body temps. It's one thing that has limited fungal infections in humans so far.Carnivorous mushrooms?
We're doomed!!!
The headline comparing this to "nerve gas" is exaggerated. It's really only toxic to microscopic organisms, where it creates a surfactant interface between the Oxygen and the carbon chain, and in high enough concentration it can dissolve the membranes of microscopic organisms. These types of ketones are mostly harmless to organisms with skin. In fact, these ketones actually tend to contribute to the slight "fruity/floral accents" for a wide variety of plants.Interesting that such a simple molecule could be so toxic. It's just a straight-chain octane with an oxygen.
https://pubchem.ncbi.nlm.nih.gov/compound/3-OctanoneView attachment 50470
The team also induced thousands of random genetic mutations in the fungus. Those mutants that didn't develop toxocysts on their hyphae were no longer toxic to the nematode Caenorhabditis elegans.
If you happen to be cold-blooded, fungi can be a pretty serious problem. They just don't tend to do so well at ~98°F.Carnivorous fungi are two words I did never imagine that I would see used together.
Humans handle high levels of simple ketones just fine. Nail polish removal is often acetone(2-propanone), and it's safe to soak your fingers in it. Methyl ethyl ketone(2-butanone) is used similarly. It's like how hand sanitizer uses ethanol to kill bacteria, while humans drink it recreationally.The headline comparing this to "nerve gas" is exaggerated. It's really only toxic to microscopic organisms, where it creates a surfactant interface between the Oxygen and the carbon chain, and in high enough concentration it can dissolve the membranes of microscopic organisms. These types of ketones are mostly harmless to organisms with skin. In fact, these ketones actually tend to contribute to the slight "fruity/floral accents" for a wide variety of plants.
That's the same reason just put two different ways.Loved the article. The article mentions in one place that the fungus may be carnivorous because rotting wood is low in protein and then ends with the speculated reason being that rotting wood is low in nitrogen. Are there theories for both those reasons: low nitrogen and low protein?
The two statements are saying the same thing differently. Nitrogen is needed by organisms because it is a primary component of amino acid (“amino” being cognate with “ammonia,” which is a compound of nitrogen and hydrogen), which in turn make up protein.Loved the article. The article mentions in one place that the fungus may be carnivorous because rotting wood is low in protein and then ends with the speculated reason being that rotting wood is low in nitrogen. Are there theories for both those reasons: low nitrogen and low protein?
